Table Of Content

The studies varied considerably in terms of number of steps and number of participants (Table 3). It may be relevant to question whether there is a minimum number of steps required for the trial to be classed as a stepped wedge. In two of these studies [14, 17] participants are randomised to two cohorts, with one cohort receiving the intervention while the other cohort served as the control.
Current Developments in Cluster Randomised Trials and Stepped Wedge Designs - Pragmatic Clinical Trials Unit - QMUL
Current Developments in Cluster Randomised Trials and Stepped Wedge Designs - Pragmatic Clinical Trials Unit.
Posted: Mon, 25 Jul 2022 07:00:00 GMT [source]
Challenges
Effect of a Multifaceted Intervention on the Incidence of Hemodialysis Catheter Dysfunction in a National Stepped ... - ScienceDirect.com
Effect of a Multifaceted Intervention on the Incidence of Hemodialysis Catheter Dysfunction in a National Stepped ....
Posted: Thu, 27 Jul 2023 19:57:28 GMT [source]
With a full-grain leather upper, molded EVA sole and oversized scalloped treads, Sorel’s Kinetic wedge sandal is sporty and stylish. The adjustable hook-and-loop ankle strap makes it easy to customize the fit and the 1-inch heel-to-toe rise gives you enough of a boost without leaving you unsupported so you can walk comfortably in this pair. Saul Bass (1920 – 1996) was instrumental in create a new American Minimalism in modern graphic design. With economy of color and utilizing almost rough, elemental forms his designs have become icons of the era, with many of his logo and corporate identity works still in use today. Perhaps John Entenza, editor of Arts & Architecture magazine who spearheaded the Case Study House program (and himself lived in CSH #9), simply lost count when assigning the commissions.
Planning and Analysis of Trials Using a Stepped Wedge Design
We have identified a wide range of SWT designs, classified in terms of how individuals experience control and/or intervention conditions, and how outcome measurements are obtained. We have also offered guidance for the choice of key design features that are more directly under the control of researchers, such as the randomisation method, the number of steps, and the step length. This work describing the range of designs contrasts with the previous SWT design literature, which has generally focussed on designs where data are collected cross-sectionally at discrete time points. As shown in our review, these constitute only a minority of recently conducted trials [1]. The review in this article provides an accessible entry point to the recent developments in methods for SW-CRTs, particularly on available tools to assist design and analysis.
Methods: Mind the Gap
In many contexts, and in most trials in our review, it may be considered that since participants have been exposed to the control condition for a long time before the trial, they are likely to be stable in relation to the primary outcome. In this case it is unlikely that exposure to the control condition or duration on response to intervention will be an issue, so the standard analysis methods and interpretation will apply. Even if participants are stable at the start of the SWT, if the primary outcome is time-to-event (or rate of detection of a condition) then designs such as the closed cohort will always be susceptible to survivor bias.
Stepped Wedge Cluster Randomized Designs for Disease Prevention Research
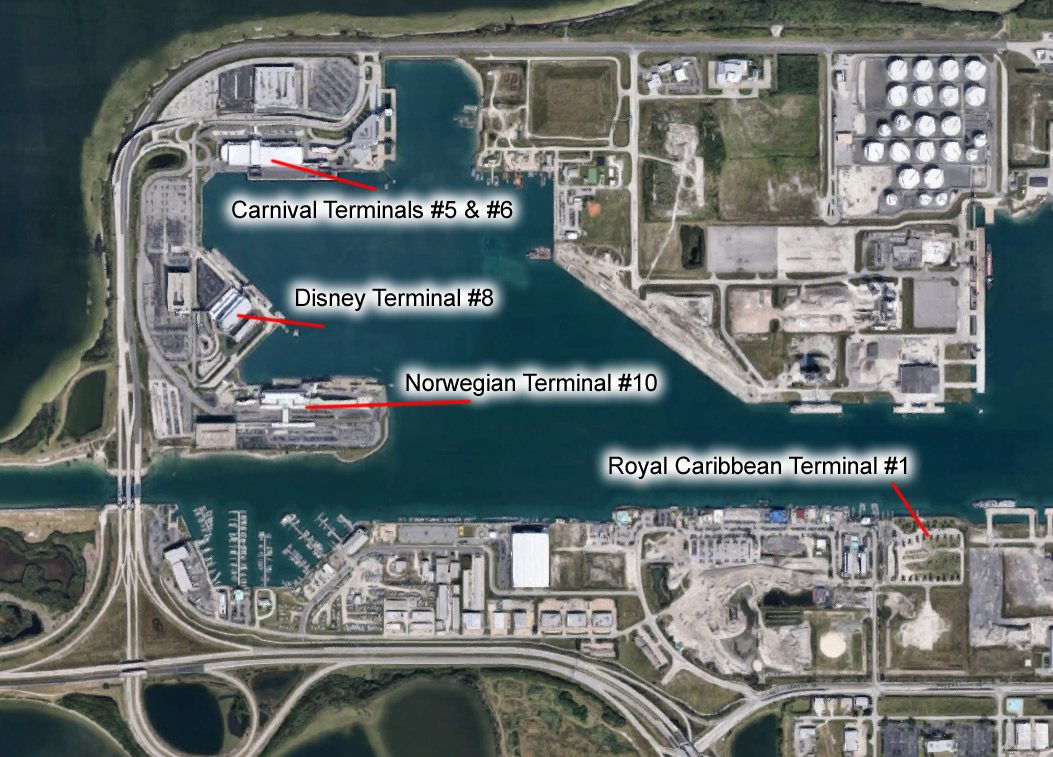
The potential benefits of employing a stepped wedge design can be illustrated by considering the £20 m evaluation of the Sure Start programme in the UK [8]. The Department for Education and Skills ruled out a cluster trial where the deprived areas identified as in need of Sure Start would be randomised to either receive the intervention or act as controls, since to intervene in some areas but not in others was judged unacceptable. The evaluation has instead used a non-randomised control group, consisting of 50 "Sure Start-to-be" communities, compared to the 260 Sure Start intervention communities. However the local programmes, evaluated by Belskey et al. [9] were in fact introduced in six waves between 1999 and 2003 [10]. This implementation strategy actually provided an excellent opportunity for a stepped wedge study design, which would have met both ethical and scientific imperatives.
Study Design and Sample
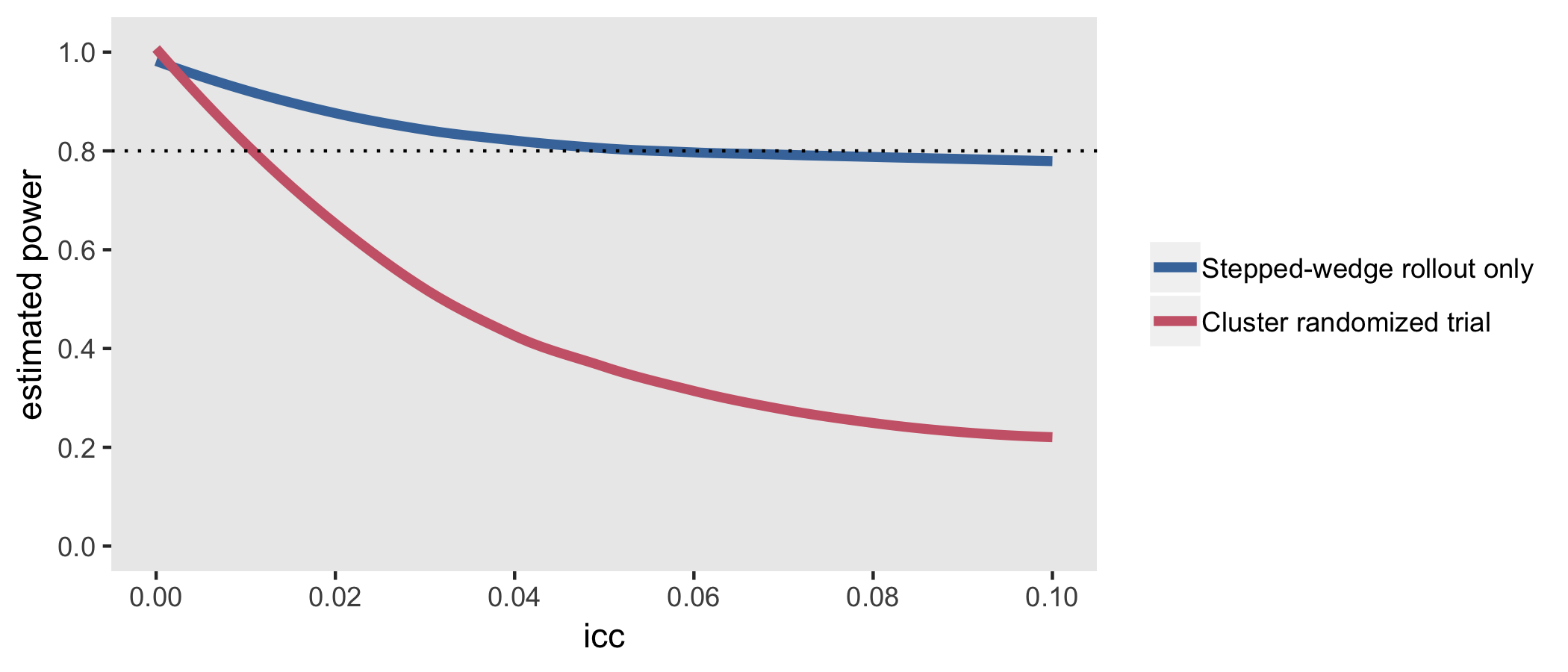
In SW-CRT design, more clusters receive the intervention toward the end of the study than in its early stages. Thus, the effect of the intervention might be confounded by an underlying temporal trend, especially if an outcome is already expected to improve over time. This consideration is particularly challenging for large-scale primary care studies, in which there can be variation across sites related to the breadth of the study and recruitment delays. All interviewees reported that SW-CRTs can be highly effective for large-scale intervention implementations. Interviewees noted that if an intervention is expected to provide a benefit with minimal risk, it is “unethical not to do the intervention for all” (North Carolina cooperative). Under that shared belief, the 3 cooperatives that did not select the SW-CRT design selected the parallel CRT design or the 2×2 factorial design, which also allow for delivery of the intervention to all sites.
In a stepped wedge study, the sample size calculation is complicated by the need to allow for the confounding effect of calendar time (an issue discussed later in more detail), and this means that the standard design effect is no longer applicable. Compared with a simple parallel study, where no such confounding occurs, the time effect tends to degrade the precision of the study and increase the sample size needed to achieve adequate power. On the other hand, each cluster in a stepped wedge study contributes both exposed and unexposed observations, and so, to some extent, acts as its own control.
The stepped wedge trial design: a systematic review
Each interview had 1 primary interviewer (A.M.N. [female] or M.P.B. [male]), with the other present to ask clarifying questions. Interviews were conducted by telephone or video conferencing, lasted approximately 30 minutes, and were audiorecorded with permission. As noted above, it is almost impossible to blind participants or those involved in delivering the intervention from being aware of whether a participant is currently in the control or intervention section of the wedge.
Method of Analysis—Marginal Models
Referring back to our three main designs, we see that in this typology a closed cohort is typically T1/D1/M1. An open cohort with repeated cross-sectional sampling for outcome measurement is T2/D2/M3. The continuous recruitment short exposure period design is T4/D3 with either M4, M2, or M5. Given the timing and duration of exposure, there are often multiple choices of outcome measure types and data collection methods, but some would be inefficient or inappropriate.
Discounting sites’ preferences put the cooperatives at risk of losing sites; however, accounting for preferences subjected the study to unequal distribution of site characteristics (eg, sites that start early differ from those that start late). Interview guides (Supplemental Appendix 1) asked each grantee to share what worked, what challenges they experienced with their study design, and lessons learned from using their design. Participants from ESCALATES were asked to reflect on experiences harmonizing data from the different study designs across cooperatives.
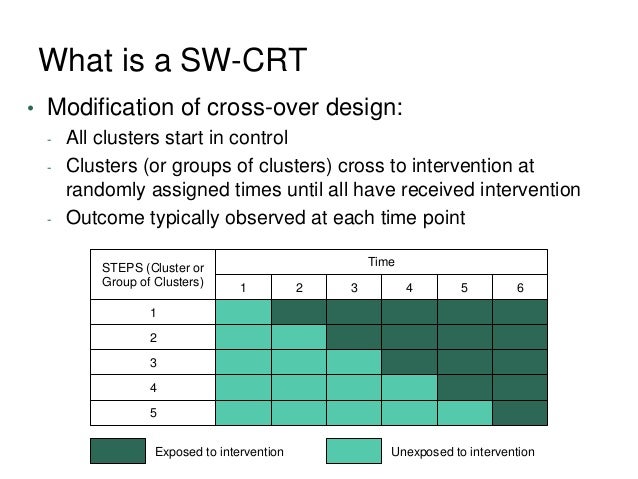
Hence most of the period of data collection does not relate to the rollout period, which provides the most direct information concerning the effect of the intervention. For all SWTs data will be collected during a rollout period (j), in which groups of clusters are crossing over from the control condition (often standard care or policy) to the intervention condition. At any one time during this period, some clusters are allocated to the intervention condition while others are not. In SWTs there may also be periods of data collection before the rollout period (g) and/or after the rollout period (i). In some trials, individuals are exposed to the control and/or intervention condition within the trial, but are then measured later after a (potentially long) follow-up period.